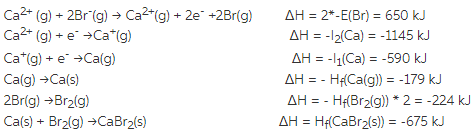
In this simple view, appropriate number of cations and anions come together to form a solid. But for simplicity, let us consider the ionic solids as a collection of positive and negative ions. There are many other factors to be considered such as covalent character and electron-electron interactions in ionic solids. Lattice Energy is Related to Crystal Structure The lattice energies for NaCl most often quoted in other texts is about 765 kJ/mol.Ĭompare with the method shown below Na(s) + 0.5 Cl 2(l) ® NaCl(s) Data from various sources differ slightly, and so is the result. The value calculated for U depends on the data used. Hf of NaCl = -411 (Enthalpy of formation) IP of Na(g) = 496 (Ionization potential or energy)ĮA of Cl(g) = -349 (Electron affinity of Cl) Hsub of Na = 108 kJ/mol (Heat of sublimation)ĭ of Cl2 = 244 (Bond dissociation energy) For a quick review, the following is an example that illustrate the estimate of the energy of crystallization of NaCl. How is lattice energy estimated using Born-Haber cycle?Įstimating lattice energy using the Born-Haber cycle has been discussed in Ionic Solids. The solids consists of divalent ions have much larger lattice energies than solids with monovalent ions. 
As the ionic radii of either the cation or anion increase, the lattice energies decrease.The larger negative value we have for the lattice energy, the more energy released when the lattice was formed, and thus the stronger the lattice structure and the higher the bond order.\): #DeltaH_"lattice"("SrS") = ?# (cannot find) #"CsI"# ( smallest charge magnitude less similar ionic radii than #"KBr"# larger ionic radii than #"KBr"# as a #pm1# ionic compound).#"KBr"# ( smallest charge magnitude most similar ionic radii smallest ionic radii as a #pm1# ionic compound).#"SrS"# ( highest charge magnitude large difference in ionic radii).So, we expect the highest lattice energy to least lattice energy to be: The above three methods of comparison are all ordered by their effect on decreasing bond strength. The charge magnitude affects the lattice energy the most by far, followed by the actual ionic radii. #"184 pm"#, #Deltar_("ionic") = "52 pm"#)Īnd finally, in order of largest to smallest charge magnitude, we have: In order of most similar to least similar ionic radii, we have: In order of smallest to largest ionic radii, we have: Now consider these ions on the periodic table: Increasing bond order is directly proportional to increasing lattice energy. The larger the charge magnitudes of the cation and anion, the stronger the bond.The smaller the difference in ionic radii between the cation and anion, the stronger the bond.The smaller the ionic radii of the cation and anion, the stronger the bond.When considering ionic compounds, we expect the following typical trends: Well, they're all ionic compounds, so the only practical way to obtain the "bond order" is through the lattice energies.īond order, qualitatively speaking, is proportional to the bond strength.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
